- Presentation
- DIVAT in 1 clic
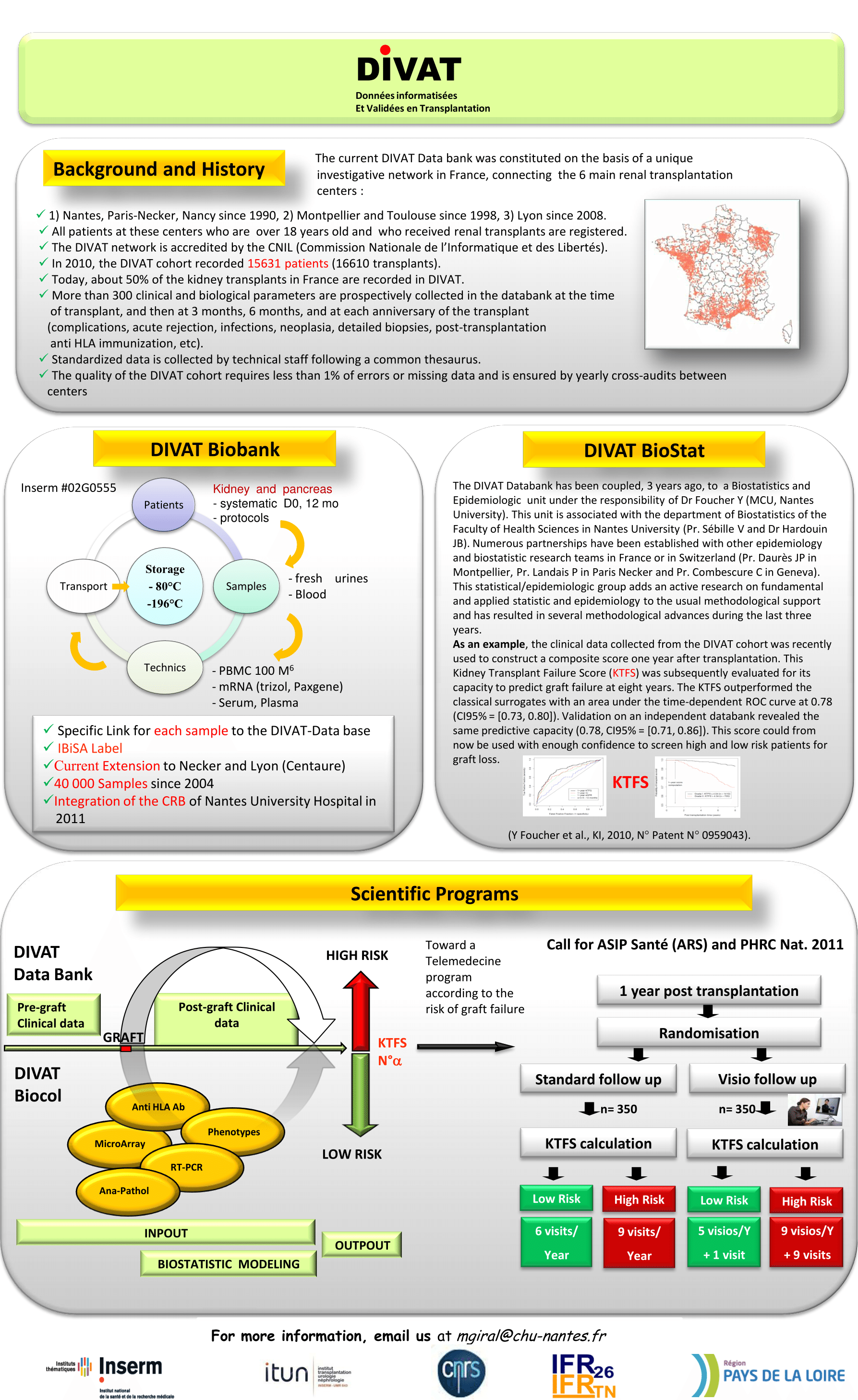
- DIVAT in figures
The Immunology and nephrology department of the Nantes University Hospital has developed a sofware called DIVAT (standing for computerized and validated data in transplantation = Données Informatisées et VAlidées en Transplantation) allowing to set up a computer database which includes the monitoring of medical records for kidney and/or pancreas transplant recipients and a software allowing data retrieval from the database for epidemiological analysis.
Nantes University Hospital has settled in the centers of Nancy, Montpellier, Toulouse, Necker (Paris), Lyon, Saint-Louis (Paris) and Nice the communication software via the Internet. Data acquisition and/or retrieval is made possible thanks to this software thus allowing data homogeneity between each network center.
The DIVAT cohort is listed on the online catalogue of the principal French health databases of the "Epidemiology - France" Portal initiated by the Alliance Nationale pour les Sciences de la Vie et de la Santé. Read more.
The eight centers established between them collaboration in order to be able
- To collect and exchange information,
- To conduct clinical and epidemiological studies all together (increase of the statistical power and better representative population),
- To communicate and exchange data and results with other academic researchers (not included in the DIVAT network),
- To construct partnership with pharmaceutical industries,
- To create a biocollection database linked to clinical data for centers of Nantes, Necker and Lyon.
The eight centers decided the creation of a centralized database with a web-network
- A central database is carried out in Nantes with an Oracle server (IDBC/A2Com).
- Data are collected by Clinical Research Assistants (CRA) according to a directory which is updated each year.
- The system is validated by an annual audit between centers. A CRA who does not belong to the audited centre is appointed by the network and carries out the audit. This audit focus on (i) quality for database collection and recipient follow-up and quality of data collected of donor and (ii) comprehensiveness for data collection. Automatic controls regarding accuracy are also generated each week for each centre.
- The central database (Information Commissioner’s Office data privacy declaration in France n°914184, CNIL agreement) brings together all data from each participating centre. Queries can be carried out directly from the Internet in a secure way.
The central database allows
- Having secure data,
- Maintenance and global updating,
- Internal messaging between all centres,
- Distant remote/control for online support.
=> Data have been collected since 1990 for Nantes, Nancy and Necker, 1994 for Montpellier, 1997 for Toulouse, 2006 for Lyon and 2013 for Saint-Louis and Nice.
Participants
Other centers can join DIVAT network. The center candidature should receive full agreement of the founding centers. The decision will be considered as final once the first quality audit is done.
Conformity of the DIVAT Network to the legal requirements
The Commission Nationale Informatique et Libertés (CNIL, Information Commissioner’s Office for data privacy declaration) has given a favorable opinion (09-17-2004, number n°891735, Réseau DIVAT: 10.16.618). The Network commits to respect the requirements and procedures of this commission.
A preliminary consent from patients regarding access to their medical record, and regarding collection and data processing, must be obtained as well as a specific consent for connected biocollection. The clinical research assistant will make sure that the informed consent has been understood and signed by the patient, before recording patients data in the database. Data from the patient medical record will be made anonymous when processing to the data retrieval. A computer access key (unique per patient) is systematically created in the database for each patient, allowing if necessary to have exchange data while keeping patient information anonymous.
Creation of the database on each site
The centers must create their own base respecting the following methods:
- A clinical research assistant or a nurse completes the patient identity, collects medical histories, basic data, immunological data, clinical and treatment data with the associated follow-up, anatomopathological and ultrasound data of the recipient. The CRA or nurse also collects donor parameters which are those registered by the "Agence de Biomédecine".
- Follow-up parameters are then collected at 3 months, 6 months, 1 year and then every year. Parameters are collected by the CRA from the patient's record and input in the computer database.
- All the items must be collected according to the reference procedure settled between the six centers (Thesaurus). All items are compulsory.
- Each modification is linked to a date of last modification of the base. All modifications of the database and/or retrieval database are registered in a document which is updated twice a year by the CRA located in Nantes. These documents are archived in Nantes and can be shared with other centers by a secure Internet access.
- The percentage of data filled out can be computerized since the beginning of the database or every year. This filling out parameters allows the missing data to be evaluated.
- An audit will be carried out once a year by a CRA who does not belong to the audited centre and will be appointed by the network. Of note: no input data for two years (or more) means that we have lost sight of a patient. Lost sight patients are not included in the audit process.
- Automatic accuracy controls are carried out every week for each centre. They focus on missing data, inconsistent values, wrong dates, suspicious data and lost sight information.
Audit Procedure
This AUDIT is composed of several levels:
- One AUDIT for data capture quality for 30 files per centre, randomly chosen, among the file of living patients, who are not lost sight patients and having an operational graft function, transplanted since 2000. For each randomized file, 20 items from the compulsory basic data and 10 items from follow-up data will be controlled. 1% is the maximum error rate tolerated. Over 1%, file of the previous six months will have to be entered again.
- One AUDIT for all-inclusiveness for 10 files per centre, randomly chosen, among the file of living patients, who are not lost sight patients, having an operational graft function, transplanted during the curse of the year. For each randomized file, 3 items from compulsory data will be controlled.
- One AUDIT for missing data will be made by a comprehensive printed version of missing data.
Organization of the DIVAT network
Each center supports its own costs of creation, follow-up and development.
Property of the databases
Each center owns of its data. The database of each center is accessible with a free use and free disposal of data. Each centre can connect itself to the database from the central server and each centre can install an individual secured and secret access code for its proper database. This secret code (known only by the centre) forbids the direct access to other centres.
Each center is responsible for the quality of its data and must accept to submit itself to audits.
The database is coupled with a simple statistical module which allows recorded data to be analyzed in real time:
- Creation of samples
- Creation of subgroups
- Creation of files
- Export files from Excel into Stat View
- Kaplan Meier curves
- Calculation of means
- Comparison of means
- Calculation of Chi-square
- Creation of items
- Link with the statistic module
Collaboration between centers
Centers can collaborate between them or they can use the entire database. A centre can use the data of another centre only with the agreement of this one. This collaboration must be made official by writing a document. If the collaboration does not concern all the members of the network, the associated centres will have to inform the other centres (Form for project of collaborative study).
Studies and Publications
Scientific work can only be initiated after the first audit of quality.
1/ Studies and works carried out by one or several centers and followed by publication
- When the entire database is used for works or studies, all centres must be previously informed of the study to be considered and give a written agreement. Moreover, common studies have to be written and the protocols have to be addressed to all the centers (Form for project of collaborative study).
- Publications will be first signed by the initiating center, followed by the signature of participating centres according to the proportion of patients included in the study in decreasing order. The mention of the DIVAT quality label is compulsory in the publication.
- Agreement of all the centers has to be collected before publication. Property rights on results will follow the same rules.
- In the case of patent or commercial concerns, a written agreement will be drafted in order to specify the rights of each partner.
2/ Studies and work by center
- Centers can freely use their own database for studies and works. Preliminary information of the network members is strongly advised. Other centres can not make any objection regarding study or publication.
- Publications will be signed only by the initiating center of the study.
- Property rights on results of works will belong to the initiating center.
- Studies and publications made by new members of the network can only be published or share after a probative period of one year.
- Centres agree to thank RCAs and the Roche Laboratory in their publications.
Financial and administrative provisions
The network members will meet at least once a year, in Paris or in cities belonging to the network, in order to have an overview of the situation. The members can be represented by the scientific leader.
All communications tools can be used for discussions. However, a written report will have to be drawn up for collaborators. These documents will be saved in Nantes hospital, which is in charge of the network secretarial tasks.
Withdrawal - Cancellation
The participating centers can leave the Network with a six months notice required. Data of the leaving center will be removed from the central database.
In case of voluntary opting out from the network or not, members will keep the right of use of the DIVAT software but will not have access to future updated data.